Silicon
Name: Silicon
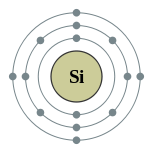
Symbol: Si
Atomic Number: 14
Atomic Mass: 28.0855 amu
Number of Protons/Electrons: 14
Number of Neutrons: 14
Number of Valence Electrons: 4
How is mass calculated: The atomic weight of silicon on the periodic table is a weighted average of the isotopic masses.
First multiply the isotopic mass by the fractional abundance to find the portion of the atomic mass from each isotope.
28Si (27.97693amu * .9221) = 25.7975amu
29Si (28.97649amu * .0470) = 1.3619amu
30Si (29.97376amu * .0309) = .92619amu
Then add the isotopic portions to find the atomic mass.
25.7975amu + 1.3619amu + .92619amu = 28.0856amu
Type of Bond Formed: Covalent bonds
Symbol: Si
Atomic Number: 14
Atomic Mass: 28.0855 amu
Number of Protons/Electrons: 14
Number of Neutrons: 14
Number of Valence Electrons: 4
How is mass calculated: The atomic weight of silicon on the periodic table is a weighted average of the isotopic masses.
First multiply the isotopic mass by the fractional abundance to find the portion of the atomic mass from each isotope.
28Si (27.97693amu * .9221) = 25.7975amu
29Si (28.97649amu * .0470) = 1.3619amu
30Si (29.97376amu * .0309) = .92619amu
Then add the isotopic portions to find the atomic mass.
25.7975amu + 1.3619amu + .92619amu = 28.0856amu
Type of Bond Formed: Covalent bonds